Article 20 (Research paper)
Zuo et al., Thermodynamic Study of Amine-Based Deep Eutectic Solvents with H2O, J. Chem. Eng. Data 2026, 71, 3, 1088–1099.
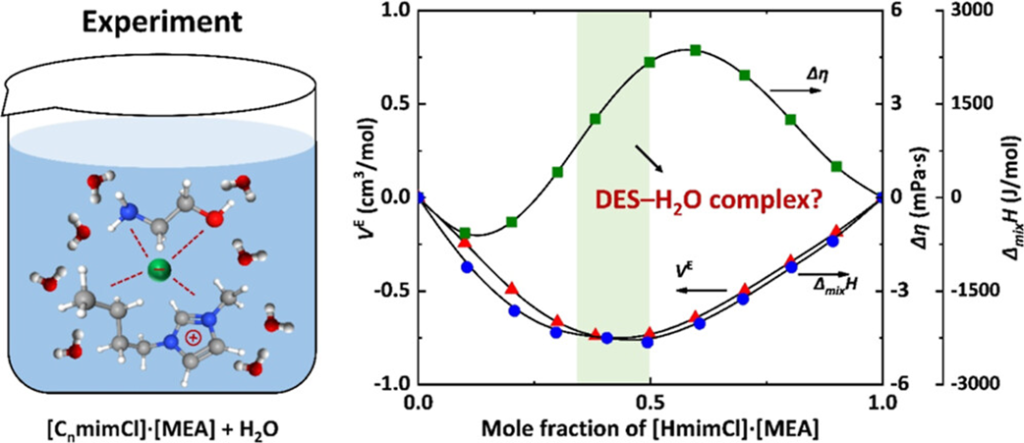
ABSTRACT: The densities and viscosities of ([CnmimCl][MEA] + H2O) systems (n = 2, 4, 6) were measured over 288.15–323.15 K, and the enthalpies of mixing were determined at 298.15 and 308.15 K. Density data showed nonmonotonic composition dependence with extrema near xDES ≈ 0.4–0.5, and negative excess molar volumes indicated enhanced molecular packing. Viscosity increased sharply at low DES content and more gradually toward pure DESs, exhibiting S-shape deviation profiles that reflect structural rearrangements in both DES-rich and H2O-rich regions. Negative enthalpies of mixing confirmed exothermic mixing due to strong DES–H2O interactions, and the data were well correlated by the NRTL model. The coincidence of extrema in density and enthalpy of mixing, together with viscosity transitions, suggests the formation of complexes among the DES constituents and H2O, probably leading to compact ternary DES-like microstructures. Further comparative analysis revealed that H2O addition reorganizes the hydrogen-bond network, forming extensive MEA–H2O associations. These findings offer molecular-level insights for the rational design of DES-based solvents for CO2 capture and related separation processes.